I have a neuromuscular disorder1, should I get a vaccine for COVID-19?
In general, yes. Most people with neuromuscular disorders (NMD)should receive the vaccine and it should protect them from the COVID-19 virus.
Clinical trial results show that COVID-19 vaccines are safe and highly effective. The side effects, for the most part, are temporary and unpleasant, but not dangerous. We do not expect the side effects to be any different for people with NMDs from everyone else.
To learn more, download our Decision Aid (pdf format).
Frequently Asked Questions (FAQ) About COVID-19 Vaccines & Neurological Disorders
Muscular Dystrophy Canada (MDC) and the Neuromuscular Disease Network for Canada (NMD4C) received numerous questions about COVID-19 vaccines and how they may uniquely affect people with neuromuscular disorders (NMD). The arrival of the first COVID-19 vaccines is a major development in the coronavirus pandemic. The rapid and successful development of not just one, but several vaccines is based on the enormous efforts of scientists, public institutions, manufacturers and volunteers world-wide.
Because the COVID-19 vaccines are still very new (~ less than 1 year of data is available), there is much to be learned specific to Canadians with neuromuscular disorders, their families, and their caregivers. Scientists are still studying the COVID-19 virus itself and how it affects people who are theoretically at-risk and vulnerable. In order for you to make informed choices for yourself and to ask questions of your own specialists, MDC asked neuromuscular specialists, respirologists, scientists, infectious disease specialists and policy-makers from across Canada to provide recommendations and information about the COVID-19 vaccines for people impacted by NMDs based on available evidence and to the best of their current knowledge/clinical expertise. Please note: these recommendations are accurate as of February 12, 2021; they should supplement, but not replace the recommendations made by your doctor or your local public health authority. Please speak to your specialist if you have any further questions.
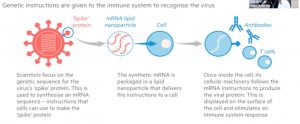
Two Health Canada approved vaccines (Pfizer-BioNTech and Moderna) are based on mRNA technology. To understand how the vaccine works, you have to first understand how the virus works. A virus contains genetic materials composed of DNA or RNA. To build what it needs, the virus will make a messenger RNA (mRNA), which is a genetic recipe for making a piece of the spikes that sit atop the coronavirus. The mRNA reads and translates information out of the nucleus into specific proteins and structures. The mRNA vaccine contains a mRNA coding for a critical piece of the virus. In the case of COVID-19 vaccine, it’s a spike protein located on the virus’ outside coat.
Our immune system will learn to attack this spike protein produced from the mRNA vaccine. Basically, when the vaccinated person comes into contact with the virus, the immune system will recognize the spike protein and launch a rapid and effective attack.
If a
vaccinated person is later exposed to the coronavirus, those antibodies should stand at the ready to attack the virus.
mRNA based vaccines work very well and have a protective effect of more than 90% in human trials. Current approved vaccines have been effective in preventing COVID-19 cases in total and in limiting severe COVID-19 cases during the trials.
There is little data available on how well they work against current (and future) new strains, but they are expected to have some protective effect against most of them (at least for now). Early research suggests that the Pfizer-BioNTech and Moderna COVID-19 vaccines can provide protection against the COVID-19 variants identified in the U.K., South Africa and Brazil.
All vaccines go through clinical trials to test safety and effectiveness:
- Careful testing. A vaccine developer generates data from studies that have been done in animals and humans, and on the manufacturing process. Health Canada experts then review the data to ensure the vaccine: is safe, works to prevent disease and/or infection and is manufactured correctly. Physicians, infectious disease specialists, microbiologists, immunologists and other experts review the clinical data. Biostatisticians review the statistics and epidemiology. Specialists review the complicated manufacturing process.
- Continuous monitoring for problems and side effects. Data available at the time of authorization will only include information on short- to medium-term side effects. To date, there have been very few reports of adverse events/negative side effects from the vaccine. The most common side effects to the vaccine are temporary (short term) and include mild pain at the vaccine injection site, fatigue, headache, muscle pain, joint pain and chills. Once a vaccine is authorized for use, the side effect monitoring continues, with systems in place to track problems or side effects that were not detected during the clinical trials. Post-market activities are required for manufacturers to follow the long-term safety of the vaccine (enhanced surveillance, international collaboration).
In addition, international regulatory bodies are collaborating to capture global safety information. If any unexpected or serious side effects are reported:
- A thorough investigation will take place
- Information will be rapidly communicated to Canadians
The Pfizer-BioNTech COVID-19 mRNA vaccine was approved in Canada on December 9th, 2020. The Moderna COVID-19 mRNA vaccine was approved on December 23rd, 2020. Overall, the safety and efficacy of the 2 vaccines are very similar.
| Manufacturer: | Pfizer-BioNTech | Moderna |
| Type: | mRNA | mRNA |
| Authorized age groups: | 16 year-old+ | 18 year-old+ |
| Dose, route and schedule: | 0.3 mL intramuscular at 0 and 21 day | 0.5 mL intramuscular at 0 and 28 day |
| Efficacy (at least 7 days after dose 2) : | 95% | 94% |
| Long-term duration : | To date, the data shows protection for 2 months after 2nd dose. Likely ‘longer protection’ but no data yet. | To date, the data shows protection for 2 months after 2nd dose. Likely ‘longer protection’ but no data yet. |
The first dose works very well for the Pfizer-BioNTech and Moderna vaccines. Approximately 10 days to 2 weeks after you get your first dose, you are starting to become protected. This has generated discussion about people ‘waiting’ to get their second dose. You should still get a second dose: it will protect you better (especially for new variant strains) and for longer. But knowing that there are significant delays with the vaccine roll-out, it is critical to at least get the first dose as it will offer some protection against the virus. For the vaccines available in Canada, you will need two doses and there is data to show the second dose of the vaccine could be delayed to 42 days.
Not to our knowledge. The clinical trials were performed in individuals from age 12 to 85 years old and none had reported neuromuscular disorders. There is no published data reporting the efficacy or side-effects of vaccines on people with neuromuscular disorders. However, the way the vaccines work does not suggest that people with neuromuscular disorders might be at increased risk of side-effects or at lower efficacy if the neuromuscular disorder does not involve the immune system.
The expectation is that the chance of spreading or passing on the virus is reduced, but not fully eliminated when you are vaccinated against COVID-19.
Yes, COVID-19 and the flu (influenza) are two different viruses. You will also need to receive a flu (influenza) vaccine every year. Just like with the seasonal flu (influenza), current research is underway to assess if repeat vaccinations for COVID-19 and new variants may be required every year. This might be combined in one shot (flu and COVID-19) in the future.
Currently public health plans are looking at annual vaccinations (once per year). It is possible that the protection weakens over time and booster injection may be required to maintain full protection for longer. The vaccine can certainly provide protection for at least two months, according to the current available data. It still remains to be seen what the long-term immune response will be after vaccination.
According to the Infectious Disease Committee of the Society of Obstetricians and Gynaecologists of Canada, “women who are pregnant or breastfeeding should be offered vaccination at anytime if they are eligible and no contraindications exist. This decision is based the women’s personal values and an understanding that the risk of infection and/or morbidity from COVID-19 outweighs the theorized and undescribed risk of being vaccinated during pregnancy or while breastfeeding. Women should not be precluded from vaccination based on pregnancy status or breastfeeding.”
In terms of Myasthenia gravis (MG), adults with MG should get vaccinated. Generally, we anticipate that the risks associated with the COVID-19 vaccine will be relatively small compared to the risk of contracting the disease itself. Persons receiving monoclonal antibody therapy (such as rituximab or eculizumab) for MG should discuss with their neuromuscular specialists how to time the COVID-19 vaccine so the vaccine is most effective. If you are pregnant and have MG, it is best to consult with your neuromuscular specialist.
Yes. The Pfizer-BioNTech and Moderna vaccines are compatible with these therapies. While the vaccines consist of mRNA and the antisense oligonucleotides in some of the genetic therapies act on RNA, the sequences are different and would not interfere. Some gene therapies are based on adeno-associated viruses (AAV), but that is a different virus that shares little similarity with coronavirus or the vaccines. Some vaccines (Astra Zeneca and others) use adenovirus, but this a completely different virus from AAV despite the similar name.
If you are involved in a clinical trial, you should discuss the vaccine with the study team, to determine whether/when a vaccine is permitted.
Generally, the side effects should not be different or more in people who are immunosuppressed. The main risk is that the vaccine doesn’t have its intended effect and doesn’t protect from COVID-19 virus in people who are severely immunosuppressed. While persons who have immunosuppression may not have the full benefit of protection, some protection is still better than none.
Current vaccines are not authorized for use in children/youth under age 16 (18) years. There is currently not enough data available for individuals under the age of 16 years. However, manufacturers are starting to enroll children into the clinical trials.
Most likely, both the Pfizer and Moderna vaccine will also be effective in children younger than 16 or 18 years old. Since they have not been tested in these age groups, these vaccines were not approved in Canada below these age cut-offs. Clinical trials in children are underway that might change this indication.
The deltoid muscle (muscle at the top of the arm at the shoulder) is most used and preferred. For people affected by NMDs, the deltoid muscle usually does not have severe weakness or atrophy (wasting), but sometimes this can occur.
If there are reasons why the deltoid muscle is not appropriate (e.g., severely wasted/muscle loss), then another muscle can be targeted. This should not make a difference for the protective effect of the vaccine. Only one vaccine maker, Pfizer, has indicated that they do not recommend injection into any other muscle.
Vaccines cannot cause the COVID-19 virus because they do not contain the SARS-CoV-2 virus. However, people who have been in contact with the virus in the days preceding their vaccination or in the 14 days following vaccination could still develop COVID-19 virus. As the vaccines are not 100% effective, some people who are vaccinated could still get infected with COVID-19.
Minor side effects with the vaccine that typically go away after a few days have been reported. The most common side effects reported were pain, redness and/or swelling where the vaccine was injected, fatigue, headache, muscle aches, chills, joint aches and fever. Approximately 4 persons in 100,000 can have a severe allergic reaction after receiving this vaccine. The frequency of this reaction is higher than usually expected after a vaccine, but it is still very rare.
Nature and frequency of known reactions to these vaccines
| Frequency | Known reactions to these vaccines |
| In most cases (over 50% of people) | Pain in the muscle at the injection site |
| Very often (under 50% of people) | • Headache, fatigue, fever or shivering • Joint pain • Muscle soreness • Diarrhea, vomiting • Swollen armpit lymph nodes |
| Often (less than 10% of people) | Redness, swelling at the injection site |
| Rarely (less than 1 person in 1,000) | Facial swelling |
Of the 90 adverse events reported to Health Canada by Jan 15/2021, none of the effects were respiratory in nature.
In general, most people with NMDs should receive the vaccine and it should protect them from severe COVID-19. In other words, individuals with NMDs should be encouraged to receive COVID-19 vaccines because the risk of COVID-19 infections likely outweighs the potential risks of the vaccine.
Even people with NMDs on immunosuppressants should receive the vaccine when offered. For extremely rare cases where inflammatory nerve disease [such as Guillain-Barre syndrome (GBS) and chronic inflammatory demyelinating polyneuropathy (CIDP)], may have been triggered by an earlier vaccination (extremely rare side effect), please consult with your neuromuscular specialist.
Patients on deflazacort should receive the vaccine. The Pfizer vaccine is currently approved in Canada from age 16 years and up. There is more experience with vaccines against other bacteria and viruses (such as pneumococcus and influenza), which are given safely in people on deflazacort. Usually, the immune system is good enough to mount a protective response.
Yes. While there is currently no data regarding safety or efficacy of COVID-19 vaccines used in persons with SMA receiving nusinersen therapy, we do not advise that treatment should be stopped. Consult with your neuromuscular specialist about the timing of vaccination and nusinersen therapy.
Yes, in principle. There is currently a lower age limit for the vaccine (Pfizer age 16 years, Moderna age 18 years).
Same precautions, contraindications and effects as for the general population. There is no reason to think that it would make CMT worse.
Yes, the vaccine will work despite the RNA issue (not all the RNA is affected by the disease). In DM1, an expanded CTG repeat is leading to a toxic RNA effect. This expanded repeat is not expected to interfere with the vaccine’s mRNA. Moreover, the expanded repeat RNA leads to a disturbance of the processing of other RNA molecules, called splicing. The mRNA in the vaccine does not need to be spliced and should not be affected by the splicing defect.
Yes, definitely once the vaccine is approved for your child’s age by Health Canada Clinical trials are underway for children 6-18 years of age. However, the vaccine is NOT yet approved for individuals less than 16/18 years of age yet. We do not yet have a clear timeline for when the vaccines will be available for children of all ages in Canada.
Yes. Generally, we anticipate that the risks associated with the COVID-19 vaccine will be relatively small compared to the risk of contracting COVID-19 itself. Patients should discuss vaccination timing with their neuromuscular specialist.
Yes. Also, it should be noted that no instances of CIDP or MMN were seen during clinical trials of the two vaccines. If you are receiving treatment with IVIg or other immune suppressing medication, you should discuss your case with your treating physician (as per discussion above for myasthenia gravis).
Yes. If you are receiving treatment with IVIg or other immune suppressing medication, you should discuss your case with your treating physician (as per discussion above for myasthenia gravis).
Yes. Persons with a history of GBS and autoimmune conditions may receive COVID-19 mRNA vaccines unless they have other contraindications to vaccination.
No, not likely. No instances of GBS were seen during clinical trials of the two vaccines and no published studies suggest any cause for concern.
Questions About Vaccine Priortization
The prioritization comes from the National Advisory Committee on Immunization (NACI). This is an expert group that looks at the clinical data and they make suggestions (at a national level) on who in general should get the vaccine. The NACI provides guidance to the Public Health Agency of Canada on specific populations who should receive the vaccine first. However, it is up to every province to determine their own sequence of priorities, based on their population and needs. Therefore, there may be some differences between provinces on the order of priorities. Currently, front-line healthcare workers and people living in long-term care facilities are prioritized. As vaccine supply increases, NACI will recommend other groups to be prioritized. But ultimately, it will be up to each province to take the NACI guidance and figure out who they will prioritize in each phase of the vaccine roll out.
From a clinical perspective, persons with neuromuscular disorders can be very different – some do relatively well, some are fully independent, some are going to work. Others might have significant difficulty with their respiration, or they might be on a ventilator and may be at high risk for pneumonia.
Certain people who have severe NMDs with compromised lungs or respiration should be high-priority.
Not all persons with NMDs are in a highly vulnerable situation. The reason for some people getting the vaccine before others is to try and prevent the most vulnerable people from getting the disease. This includes people who would likely get very sick if they were infected. Additionally, health workers such as those working with the most vulnerable in ICUs, emergency departments and long-term care are exposed repeatedly to persons with COVID-19, and if they were to get sick, our hospital and long-term care centres would not be able to take care of people.
Individuals considered at high or very high risk for COVID-19 are people with severe or unstable respiratory compromise; reliance on home ventilation; clinically relevant impairment of heart function; immunosuppression; or severe weakness requiring multiple caregivers or complex ongoing support in the home, community or institutional setting. If you would like to learn more about vaccine prioritization and how you can be specifically prioritized in your region, contact your Service Specialist at MDC. MDC will be able to work with you to support your local advocacy initiatives and can help with meetings and letters of support.
QUESTIONS ABOUT NMDS & COVID-19
The information outlined in the Spring Edition of the NMD4C and MDC ‘Ask the Experts’ still holds true. Learn more: https://neuromuscularnetwork.ca/news/ask-the-experts-covid-19-and-nmd/
Even though clinical observations have noted people with Myasthenia Gravis and Myotonic Dystrophy to be at particular high risk for COVID-19, we do not have the data to say 1 type of NMD is at higher risk than others. Some factors that would put individuals with NMD at higher risk include:
• Taking medications that suppresses the immune system (e.g., deflazocort)
• Respiratory complications (e.g., using a ventilator, a weak cough)
• Cardiac complications
• At risk of deteriorating or developing rhabdomyolysis during fever, fasting or infections
The Neuromuscular Working Group of the Spanish Pediatric Neurology Society recently published a study which describes the clinical characteristics and outcome of COVID-19 in children with NMDs. The most common NMD in the study were SMA type 1 and 2 (including patients undergoing treatment with nusinersen (Spinraza), risdiplam or salbutamol), and Duchenne Muscular Dystrophy (including patients undergoing treatment with deflazacort), along with several other neuromuscular conditions.
Although derived from a small sample size, the study’s conclusions suggest that the course of COVID-19 in children with NMDs may not be as severe as expected. It appears there is a protective role of young age that seems to outweigh the risk factors that are common in children with NMDs, such as a decreased respiratory capacity or a weak cough.
Viruses mutate all the time, and the COVID-19 virus has been mutating since it first latched onto humans. We predict the virus will continue to mutate. No new precautionary measures are suggested at this time: you should continue to practice good hygiene/proper and frequent hand washing; wear a mask; maintain social distancing; and get vaccinated as soon as you are eligible.
This will largely depend on the rollout of the vaccine. In the US, Dr. Fauci (National Institutes of Health NIH) said if 70 to 85% of Americans get vaccinated by the end or middle of the summer, a “degree of normality” can be expected by around the fall. However, this could be at a different time in Canada.
Acknowledgements:
Responses were provided and reviewed by Dr. Reshma Amin, Dr. Erin Beattie, Dr. Megan Bettle, Dr. Valérie Gagné-Ouellet, Dr. Hans Katzberg, Dr. Rashmi Kothary, Ms. Stacey Lintern, Dr. Hanns Lochmüller, Dr. Allison McGeer, Dr. Anne Pham-Huy, Dr. Colleen O’Connell, Dr. Homira Osman, Dr. Gerald Pfeffer, Dr. Rachel Thompson, Dr. Jodi Warman Chardon and Dr. Toshifumi Yokota.
Responses were also based on a webinar organized by NMD4C and MDC. If you have any questions, please email research at muscle.ca
(1) Muscular Dystrophies;
(2) Congenital Muscular Dystrophies;
(3) Congenital Myopathies;
(4) Auto-immune Mediated Myopathies;
(5) Metabolic Myopathies;
(6) Genetic and Immune-Mediated Neuromuscular Junction Disorders;
(7) Hereditary Peripheral Nerve Disorders (Genetic Neuropathies);
(8) Immune-Mediated Peripheral Nerve Disorders and;
(9) SMA and related lower motorneutron disorders.
