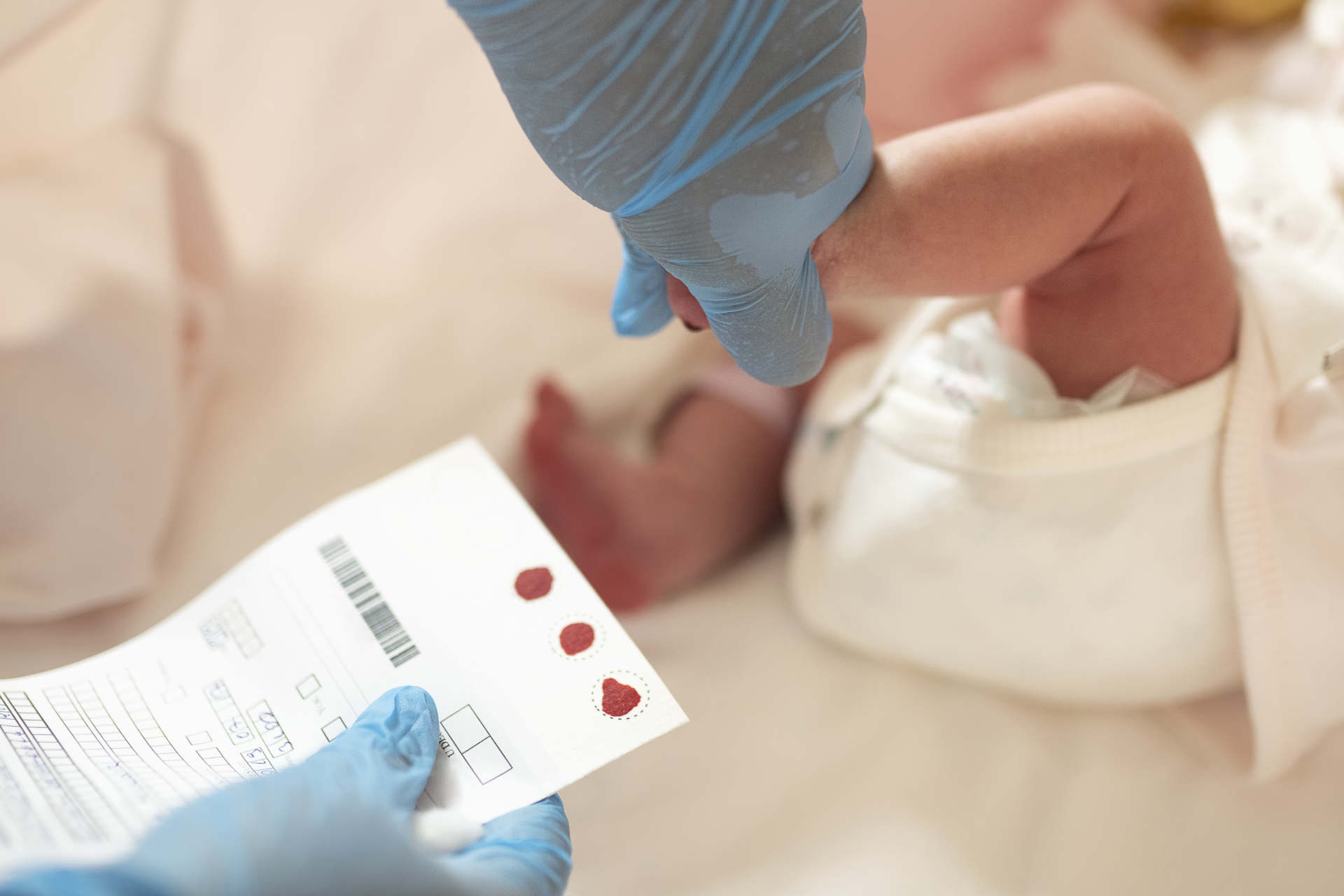
- SMA is the leading cause of genetic infant death.
- Early diagnosis is imperative to halt irreversible motor neuron loss and disease progression.
- More than $700,000 awarded to projects across three provinces.
Toronto, ON, July 21, 2021 – Muscular Dystrophy Canada (MDC) today announced funding for newborn screening projects focused on spinal muscular atrophy from Alberta, Manitoba and Quebec. These are the first applicants to be awarded funds as part of a collaboration with Novartis Pharmaceuticals Canada Inc. (Novartis). This multi-year initiative will see up to $2 million invested, as well as ongoing support for the generation of critical evidence and best-practices to help make newborn screening for SMA a national reality.
“In a neuromuscular disorder like SMA, where time is of the essence, early diagnosis and prompt access to treatments are critical to achieving the best possible outcomes. Unfortunately, this is not the reality for most Canadian children born with SMA,” said Stacey Lintern, CEO, Muscular Dystrophy Canada. “With today’s announcement and the unparalleled commitment of government leaders, Novartis, the SMA community and MDC’s dedicated Board of Directors, Fire Fighters, clients, donors and supporters, we are one step closer to ensuring all Canadian newborns are screened for SMA.”
Newborn screening is a test done for babies shortly after birth to look for treatable diseases that usually show no symptoms in the newborn period. While Ontario is currently the only province to screen for SMA, MDC’s goal is to have the disorder added to all provincial newborn screening panels across Canada.
“As the mother of an infant with SMA who was not screened at birth, I know first-hand the devastating implications of delayed diagnosis,” said Laura Van Doormaal, Newborn Screening Advisory Committee member. “Today’s announcement means that one day soon, no matter where they live all families will have the opportunity to make critical healthcare decisions and realize their child’s full developmental potential from the very beginning.”
In order to ensure an evidence-based approach to funding, MDC and Novartis commissioned a readiness assessment to evaluate the feasibility of adding SMA to all provincial and territorial screening panels. Proposals were then evaluated by an independent international peer review committee under MDC’s guidance. Funded projects include pilot programs and the mapping of post-diagnosis referral pathways. Further details on the needs and readiness assessment, project selection, peer review and community advisory committees can be found here: muscle.ca/newborn-screening
“Our collaboration with Muscular Dystrophy Canada truly exemplifies the Novartis purpose of improving and extending patients’ lives,” said Andrea Marazzi Country Head, Novartis Pharmaceuticals Canada. “We’re honoured to partner on work that will bring lasting impact to the SMA community and healthcare systems across Canada. Time to diagnosis is crucial in SMA and we applaud the first round of funding recipients and all engaged provinces for their commitment to prioritize screening at birth.”
MDC will continue to work with British Columbia, Saskatchewan and the Maritimes to accelerate newborn screening for SMA coast-to-coast. Future phases of the national collaboration will include additional project funding, evaluation of projects and knowledge transfer, and exchange with stakeholders to ensure policy adoption across all provinces and territories.
About Muscular Dystrophy Canada
Muscular Dystrophy Canada’s mission is to enhance the lives of those affected with neuromuscular disorders by continually working to provide ongoing support and resources while relentlessly searching for a cure through well-funded research. To learn more about Muscular Dystrophy Canada, please explore our website or call our toll-free number at 1-800-567-2873.
-30-
For more information:
Heather Rice
heather.rice@muscle.ca
902-440-3714